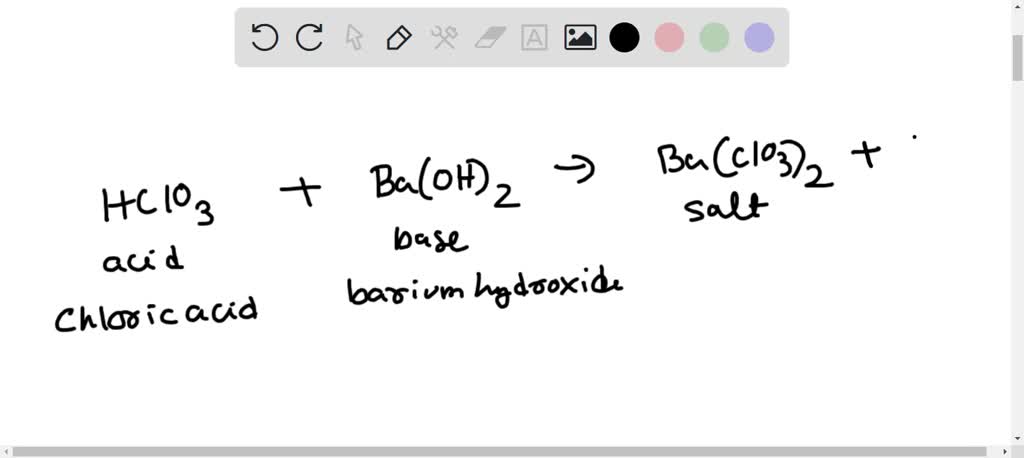
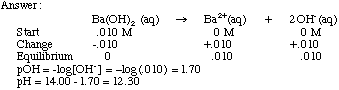Is Ba(OH)2 an Acid, Base, or Neutral? (Barium Hydroxide) | Is Ba(OH)2 an Acid, Base, or Neutral? (Barium Hydroxide) Do you want to find out if Barium Hydroxide is an acid, base,

SOLVED: A solution of Ba(OH)2 was standardized against 0.1215 g of benzoic acid with grade of primary standard, C6H5COOH (122.12 g / mol). The end point was observed after adding 43.25 mL

How to Write the Net Ionic Equation for Ba(OH)2 + H2SO4 = BaSO4 + H2O (Note: it should be 2H2O) - YouTube

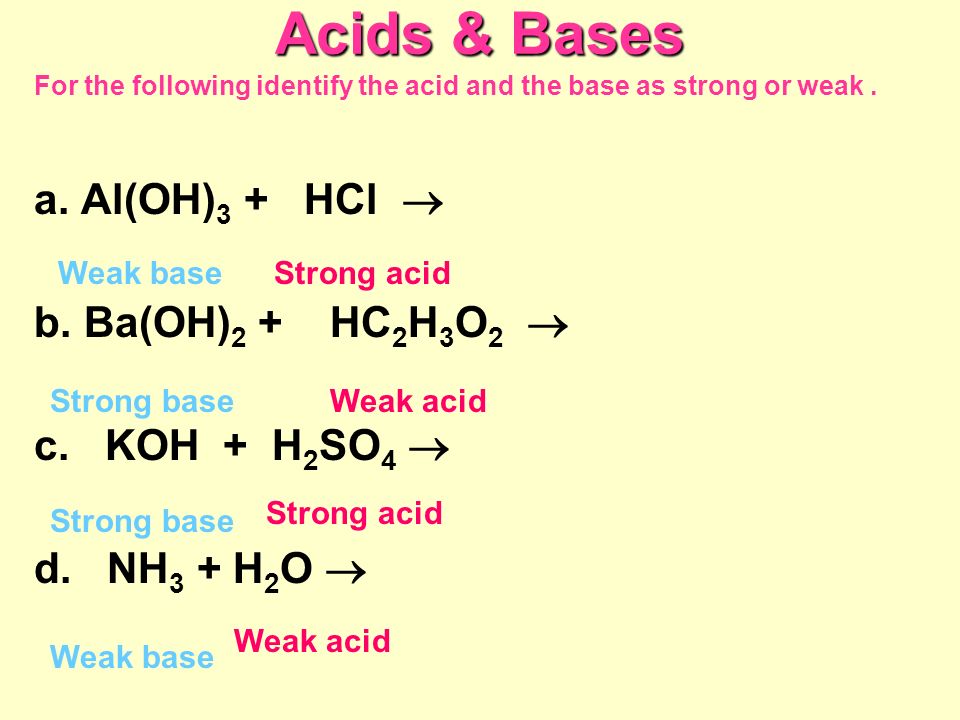
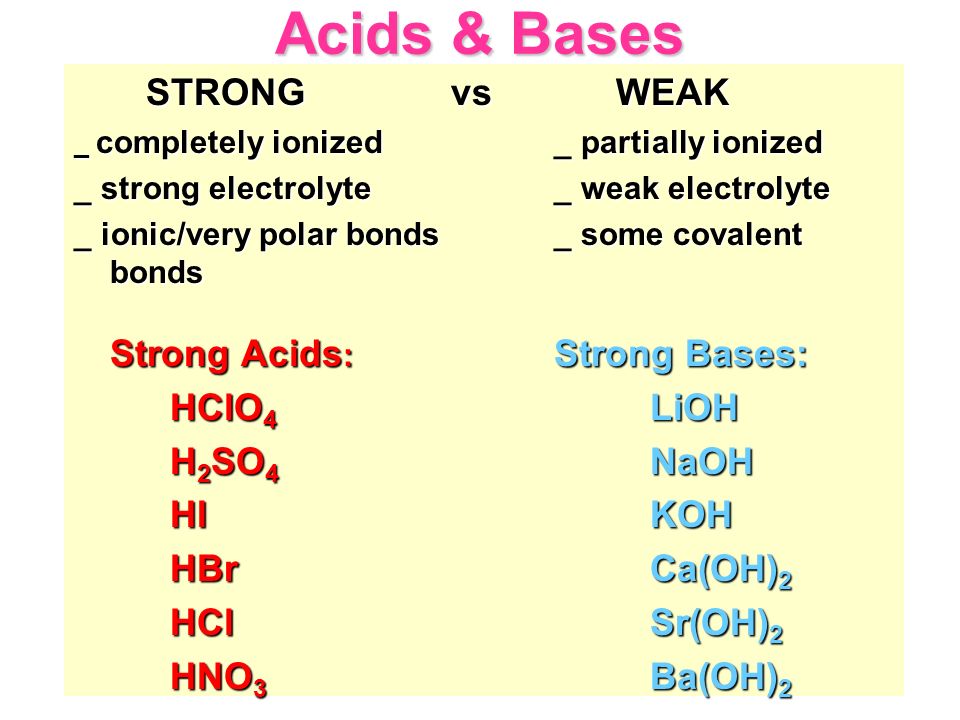
Acids & Bases Acids: acids are sour tasting Arrhenius acid Arrhenius acid: Any substance that, when dissolved in water, increases the concentration. - ppt download