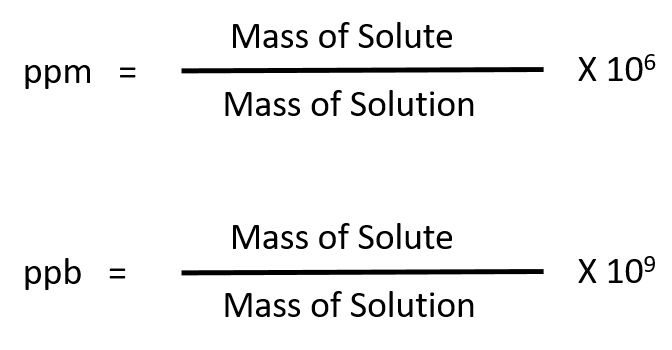
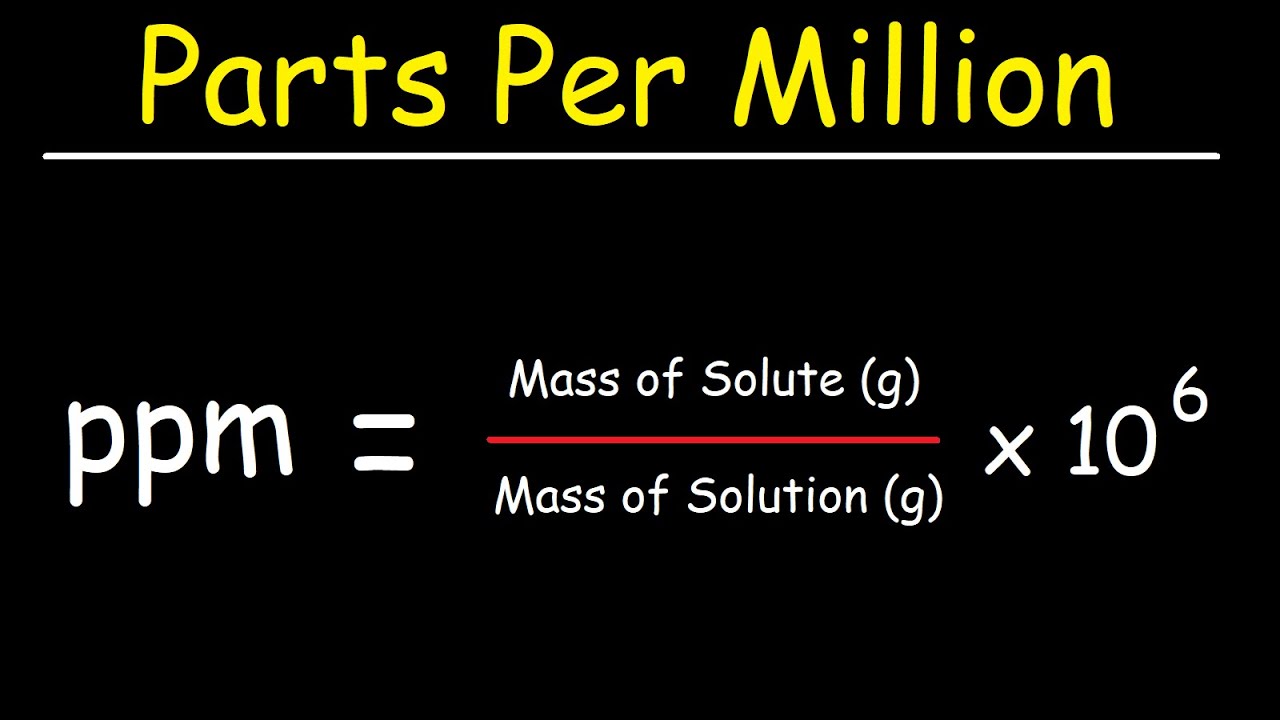Parts per Million Concentration. Parts per Million (ppm) is the ratio of the number of grams of solute for every one million grams of solution. parts. - ppt download

SOLVED: Calculate the number of mL of a 50 ppm solution needed to prepare 50 mL of a 6.4 ppm solution.